Like some of the phosphorus examples, the last two are rather complicated, with hydrogens attached to the central carbons as well as the oxygens seen in all the other cases. In addition to these examples, there are a few carbon-based oxoanions that are pretty common: carbonate (CO 3 2 -), formate (HCO 2 -) and acetate (CH 3CO 2 -). However, none of those names would signal to us that hydrogen atoms are also attached to the central atom, in addition to the oxygens, but that's what we have in phosphite and hypophosphite. The prefix per- always means an extra oxygen (beyond what we see in the -ate ion), whereas the prefix hypo- means even fewer oxygens than in the -ite ion. The ending -ate always means more oxygens than -ite, although neither one corresponds to a specific number of oxygen atoms. 
Then, it becomes clear that the names offer clues to the structure. (The only exception, and it probably isn't important at this point, is persulfate it has four oxygen atoms attached to a central sulfur atom, and the fifth oxygen hangs from one of the other oxygen atoms.) The names of each of these ions seem a little random until you consider them together. For example, in chlorate, four oxygen atoms are attached to a central chlorine atom. Most Common Oxoanions seriesĪll of these anions have oxygen atoms bound to a central atom of another kind. Some of the most common oxoanions are found in the table below. That lets us distinguish chloride, Cl -, and oxide, O 2-, from chlorate, ClO 3. Because the oxoanions are very common, they have their own names. Although oxygen does exist as a simple oxide anion, O 2-, that isn't the case in these examples. Consequently, if you spot chlorine or oxygen in a structure, you can't always be sure it is acting as a simple anion.įor example, there are host of compounds that are "polyatomic ions", clusters of atoms bound tightly together that have an overall charge. However, the halogens and other elements toward the right hand side of the periodic table display a little more versatility that the alkali and alkaline earth metals. Halogens, such as fluorine and chlorine, are frequently found as halide ions, such as fluoride, F-, and chloride, Cl. Provide formulae for the following names.Ī) lithium fluoride b) sodium iodide c) potassium bromide d) magnesium chlorideĮ) calcium oxide f) beryllium sulfide g) sodium oxide h) lithium nitride Answer a:Ī similar line of reasoning works with atoms on the other side of the periodic table. Once you have identified the cation, you can quickly decide that the other atom in the formula must be the anion. As a result, both of these electrons are easily lost, and so magnesium is most often found as Mg 2 + and calcium is usually found as Ca 2 +. These metals have two electrons in the outermost shell, and there still are not enough protons in that distant nucleus to hold those electrons tightly. The reason, of course, is that these metals all have one electron in a shell that has been newly started that shell is far from the nucleus, so the electron is ot held very tightly and it is lost pretty easily.Ī similar thing is true for the alkaline earth metals such as magnesium or calcium. The alkali metals are always found as 1+ or monovalent cations, such as Na +, K +, or Li +. They are very commonly found in ionic compounds, and they are reliably found as cations in those compounds. These are the alkali and alkaline earth metals. The key is usually found in the elements that form the first two columns of the periodic table. If you didn't know that, or if for some reason the formula did not appear in that order, you should still be able to work it out based on the layout of the periodic table. How did we know which one was the cation and which was the anion? For one thing, when we write the formula of a simple ionic compound, we usually writethe elements in the same order: cation before anion. 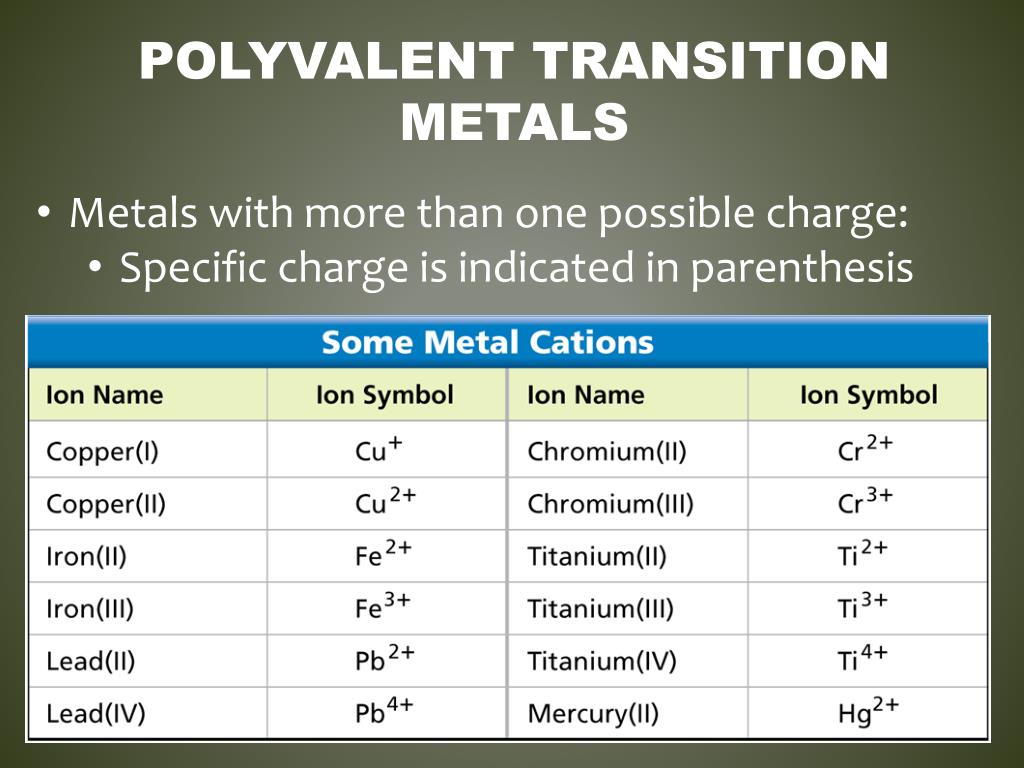
In this case, sodium is the cation and chlorine is the anion, so the name for NaCl is sodium chloride.\ 
Change the ending of the anion to -ide.Name the cation first and the anion second.We name the cation first and the anion second. We leave the name of the cation exactly as it is in the periodic table, but we change the ending of the anion's name to -ide. To name this compound systematically, we just name the two ions of which it is composed: sodium and chlorine. Take a common ionic compound such as table salt or rock salt, which has the formula NaCl. You might need to go the other direction, too if you see a formula - the ratio of atoms in a compound - you may want to be able to name the compound. Sometimes, it is useful to be able to recognize the name of a compound and know how to translate it into the structure.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
